Faster therapy timelines mean less room for variance — and higher stakes at every stage of production. CQV (Commissioning, Qualification, and Validation) pharmaceutical services ensure your systems, equipment, and processes meet Good Manufacturing Practices (GMP) and regulatory requirements while keeping production on schedule. Performance Validation (PV) delivers a comprehensive, integrated approach that’s done your way, right the first time.
No Project Too Small. No Facility Too Large.
PV partners with pharmaceutical and life sciences organizations to deliver CQV expertise that scales with your project, facility, and timeline.
- API, parenteral, and OSD manufacturing
- CDMOs and CMOs
- Late-stage clinical startups
- 3PL logistics companies
- 503A and 503B compounders
- Precursor, excipient, and packaging vendors
The CQV Pharma Process that Meets Aggressive Timelines
When you partner with us, our team becomes your team. We learn your internal processes, understand your project requirements, and provide a compliant solution that adheres to regulatory requirements and industry best practices.
- Commissioning verifies that systems are installed and operating according to design intent
- Qualification (IQ/OQ/PQ) confirms that the equipment performs as specified under real operating conditions
- Validation proves that processes consistently produce a product that meets predefined quality standards
PERFORMANCE VALIDATION’S CQV PROCESS:
Our CQV Capabilities for Optimized Pharma Products
- Risk assessments (ASTM, ICH Q9, GAMP5)
- System and component classification
- Design review support
- Regulatory gap analysis
- Critical Process Parameter (CPP)/Critical Quality Attribute (CQA) business and quality risk assessment
- Meeting facilitation and development
- Validation Master Plans and project planning
- User requirement specifications (URS) and functional specifications
- Installation Qualification/Operational Qualification/Performance Qualification (IQ/OQ/PQ) protocol development
- Standard operating procedures (SOPs) and training documentation
- Change control creation and management
- Electronic records and signatures (ER/ES) development
- Non-conformance and deviation documentation
- Commissioning execution
- Installation Qualification/Operational Qualification/Performance Qualification (IQ/OQ/PQ) testing and execution
- Factory and site acceptance testing (FAT/SAT)
- Equipment and software function validation
- Temperature and humidity mapping
- Information Technology Computer System Validation (IT-CSV), Operational Technology Computer System Validation (OT-CSV), and Robotics
- Critical airflow visualization
- On-site and remote test execution
- System turnover and formal release activities
- Vendor management, audit, and assessment
- Validation and project summary reports
- System release documentation
- Periodic review and annual audit support/audit preparation and mock audits
- Preventative maintenance program support
- Utilities and environmental monitoring trending
- Configuration and change management
- Quality Management System (QMS) development and support
- Regulatory framework transitions
Creating Better GMP Manufacturing Workflows
PV has decades of experience qualifying and validating Pilot, Active Pharmaceutical Ingredients (API), and final production lines for small and large molecules and vaccines using batch and continuous manufacturing processes. Our proactive approach surfaces risks before they become disruptions so you can keep production on schedule, documentation audit-ready, and your team focused on what matters most.
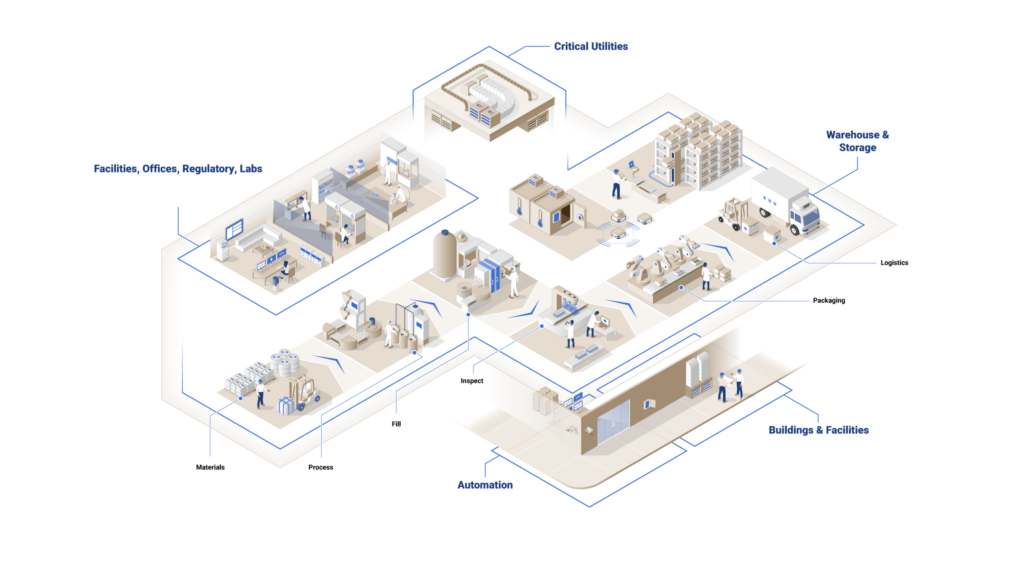
Supporting Smart Factories at Scale
Pharma 4.0 or “Smart Factories” is here to stay. PV leads top manufacturers in the deployment of automated storage and retrieval systems (ASRS) and automated guided vehicles (AGV) while supporting FDA digital transformation, lights-out automation, continuous manufacturing, and personalized medicine initiatives.

Case Study: mRNA Vaccine Rollout
Challenge
One of the largest pharmaceutical manufacturers in the country needed to produce billions of mRNA vaccines in nine months.
Approach
By collaborating with the manufacturer, PV streamlined processes by validating procedures and equipment in real time.
Solution
The pharmaceutical manufacturer saved hundreds of hours at the end of the validation process when the vaccine was ready to be distributed. The rollout of the mRNA vaccine was made possible by seamless collaborations across multiple companies and countries, making “Project Light Speed” a success.
Your Dedicated Partner for CQV Pharma Services
For over 30 years, PV has been a trusted CQV expert for FDA-regulated industries — from single system qualifications to multi-million-dollar greenfield facilities. Employee-owned since 2018, we embed quickly, work within your SOPs, and scale to match your program. Expect top-tier quality, full transparency, and a team that’s always ready to deliver.
