At a glance:
A medical device manufacturer was looking for assistance to make their Enterprise Resource Planning (ERP) system FDA compliant for an external audit coming up later in the year.
CHALLENGES:
The client did not have internal capabilities or the bandwidth to complete the validation. Their team lacked the expertise and knowledge to execute the project confidently. They were not familiar with how to approach a validation or what documentation they would need to create.

SOLUTIONS
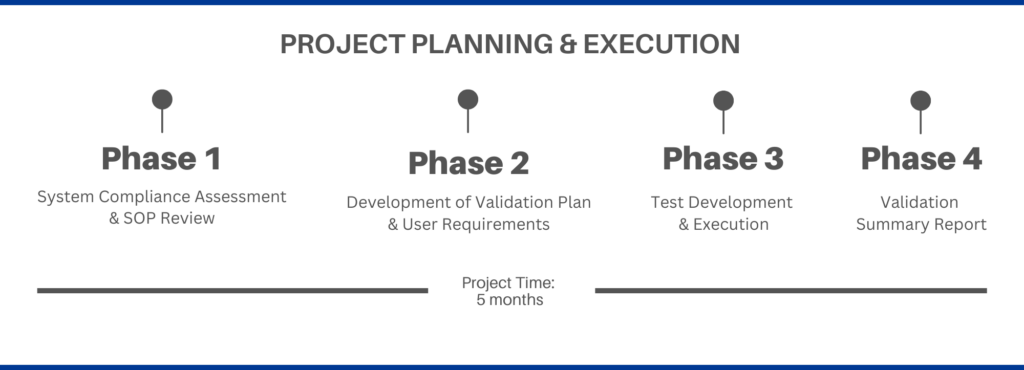
PV worked with the client to learn their Standard Operating Procedures (SOPs) to develop a plan for the approaching audit. To facilitate communication and project deadline needs, PV set specific deadlines and follow-up policies to keep the team on track and moving forward. PV collaborated with quality and business users for feedback and aided in the decision-making for document development.

PV used a risk-based approach to Computer System Validation (CSV) to determine the deliverables and level of testing needed for our client to provide the right documentation for the audit. PV’s risk-based approach is centered around our expertise in ISPE GAMP 5 and Computer Software Assurance (CSA) and many years of completing CSV projects.
An additional challenge encountered during the project was a concern that the storage drive where the client stored the validation documents needed to be validated. PV was able to work with the client on their current procedures to verify all the right procedures were in place for the storage drive; they just needed assistance with the proper documentation. The client had very specific, very detailed validation SOPs, but no validation documentation templates. PV was able to modify its templates to meet the client’s SOPs.
Another key component of the validation process is the Audit Trail. While the client did have an audit trail for the system, it was not easily accessible. PV helped define their audit trail needs to ensure regulatory compliance.
WHY PARTNER WITH PV
When choosing PV as a CSV partner, our clients can be confident that the experience and dedication our team provides is nothing less than the best. PV can lead any project from any phase of the validation process, while efficiently placing protocols and guidelines early in the planning process to meet even the strictest deadlines.
Our experience with applying CSA and GAMP 5 concepts in real-world scenarios allows our team to devise and execute the most efficient and compliant CSV solutions possible.
PROJECT SUMMARY
The PV team streamlined the risk and gap validation analysis process, reducing costs and shortening the overall project timeline. The PV subject matter experts validated the proper functions of the system according to the intended use while assisting our client with preparing and finalizing the documentation needed to be audit ready.
WE TURN CRITICAL INTO COMPLIANT
Since 1988, Performance Validation has been providing clients with expertise in the commissioning, qualification and validation service areas in the life science industries. Our team thrives on the importance of quality, integrity and teamwork, giving our clients peace of mind during every step of the project.
Download the case study below:
Dynamics 365 Computer Systems Validation
